
Legal
Indiana SB 282 Has Passed: What Med Spas Need to Know
After revisions, Indiana Senate Bill 282 has passed in both houses and has been signed by the governor. AmSpa's Alex ...

Show your committment to patient safety, legal compliance and community over competition.
AmSpa members receive preferred pricing on all AmSpa live and virtual trainings.
Get the latest news and information about safe, legal practice in medical aesthetics directly in your inbox.
Get access to med spa laws, in-person and online training and more!
Posted By Kate Harper, Tuesday, March 5, 2019
By Patrick O'Brien, J.D., Legal Coordinator for the American Med Spa Association
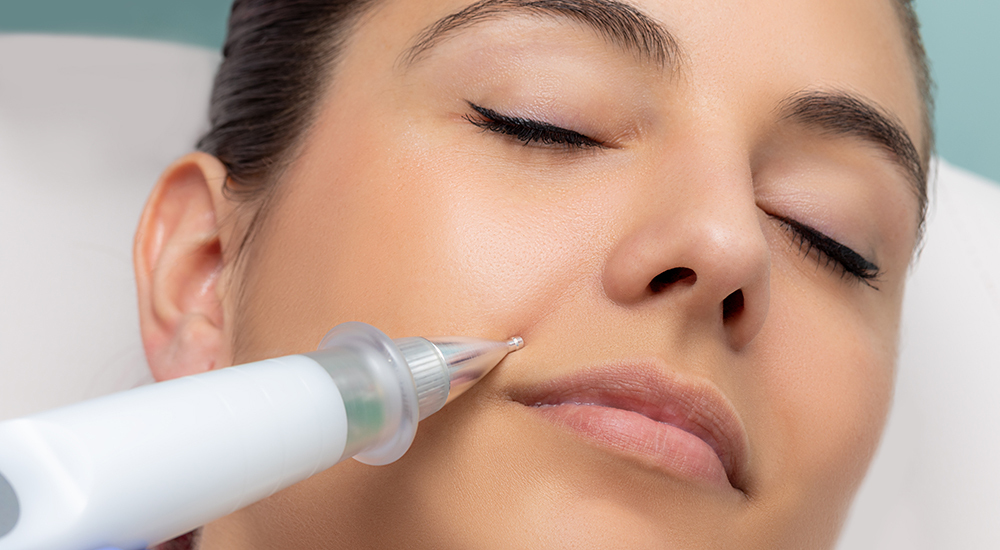
Fibroblast Plasma Therapy is a new skin tightening procedure that is generating a lot of buzz in the medical aesthetics industry. It delivers tiny arcs of plasma created by an electrical discharge via a "plasma pen" to the skin, singeing the surface and tightening the underlying tissue. Unlike a typical light or other energy based skin tightening treatment where a large area of skin is treated with each firing, a Fibroblast Plasma Therapy treatment only effects a tiny pin-point of tissue with each firing. The whole treatment is then performed by repeatedly applying the pen in grid pattern over the treatment area. It appears that, in general, there are few risks of complications with this treatment. However, there is risk of burning through part of the skin if the plasma is applied for too long or too deep.

As is typically the case when such a product emerges, we at AmSpa are getting a number of questions about who can actually perform Fibroblast Plasma Therapy procedures. The treatment appears to be very straightforward—a provider applies the device in a grid pattern to the area of the skin to the skin. Its simplicity raises an obvious question: can an esthetician or licensed vocational nurse (LVN) perform this procedure?
It's great that we get these questions because it shows that medical spa owners and operators care about remaining compliant, but because innovation moves faster than the regulation, it's sometimes difficult to determine what the answers are when new treatment emerges. However, we can use what we know about similar treatments and technologies to determine the most prudent course of action until government agencies make their rulings.
Simply put, the Fibroblast Plasma Therapy is essentially an energy device similar to other IPL, electricity, and radio frequency devices. A lot of the issues we'e addressed in recent years regarding other energy skin tightening devices are likely also going to apply to it. Every state that has looked into these other energy devices that tighten the skin has found it to be a medical treatment, so a good-faith exam must be performed before the procedure, and if a doctor is not administering the treatment him- or herself, it must be properly delegated. Unfortunately, for practices that would like to use unlicensed practitioners to perform Fibroblast Plasma Therapy procedures, this takes them out of the scopes of practice for estheticians and most LVNs.

The only conjecture we can make is that Fibroblast Plasma Therapy will likely be regulated in much the same way as other energy based skin tightening, both in terms of medical board rulings and FDA approval.
Unfortunately, since it is so new, we don't have all the answers yet, however we will stay on top of this as more states weigh in on the procedure and news develops. Look to AmSpa for more about Fibroblast Plasma Therapy treatments. Thus far, it has been safe and well received, but the industry needs to have a firmer grasp on the regulatory issues surrounding it.

To learn about legal and business best-practices to keep your med spa compliant and profitable, attend one of AmSpa's Medical Spa & Aesthetic Boot Camps and be the next med spa success story.
For legal updates and business best practices delivered straight to your inbox, subscribe to AmSpa's email newsletter. For more information on how AmSpa can help your practice operate legally and profitably, Contact Us online or call us at 312-981-0993.
Related Tags
Medical spa news, blogs and updates sent directly to your inbox.

Legal
After revisions, Indiana Senate Bill 282 has passed in both houses and has been signed by the governor. AmSpa's Alex ...

Legal
By Alex Thiersch, Founder and Chairman, AmSpaThe Indiana Senate recently passed Senate Bill 282, which includes wide-ranging medical spa regulatory ...

Legal
By Adam Reinebach, Chief Executive Officer, American Med Spa Association (AmSpa)New Jersey is at a crossroads.With the recent expiration of ...

Legal
By Madilyn Moeller, Marketing Content Coordinator, American Med Spa Association (AmSpa)A few lines in a state bill can fundamentally change ...