Featured Post
Who Can Own a Medical Spa?

Show your committment to patient safety, legal compliance and community over competition.
AmSpa members receive preferred pricing on all AmSpa live and virtual trainings.
Get the latest news and information about safe, legal practice in medical aesthetics directly in your inbox.
Get access to med spa laws, in-person and online training and more!

Marketing
By Salt MarketingJust a few months ago, JAG Medical Spa in Summerville, South Carolina was doing everything “right” on Instagram ...

Legal
After revisions, Indiana Senate Bill 282 has passed in both houses and has been signed by the governor. AmSpa's Alex ...

AmSpa Events
By Adam Reinebach, Chief Executive Officer, American Med Spa Association (AmSpa)The medspa M&A market remains active and well-capitalized as it ...

AmSpa Events
The first few years of operating a medical spa can be exciting and, at the same time, overwhelming. Nearly half ...


Marketing
By AestheticsPro For medical spa owners looking to reach their clients in the most...
Top Tags
January 24, 2023
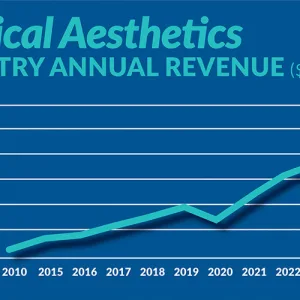
Figure displays medical aesthetics industry annual revenue ($ in billions). By Madilyn Moeller Day-to-day life...

February 24, 2021
By Bradford E. Adatto, Partner, ByrdAdattoIntravenous (IV) therapy has been used to provide nutrition and...

September 21, 2023
By Patrick O'Brien, JD, General Counsel, American Med Spa Association [AmSpa first published a...

July 19, 2023
By Patrick O'Brien, General Counsel, American Med Spa Association (AmSpa) UPDATE #2 (2/08/2024): Since...