Featured Post
Who Can Own a Medical Spa?

Show your committment to patient safety, legal compliance and community over competition.
AmSpa members receive preferred pricing on all AmSpa live and virtual trainings.
Get the latest news and information about safe, legal practice in medical aesthetics directly in your inbox.
Get access to med spa laws, in-person and online training and more!

Business
More and more people are turning to med spas for treatments that help them look good and feel even better ...

Marketing
By Ashley James, CEO, DermAesthetic Consulting GroupIf you’ve worked in the aesthetic industry for any length of time, you know ...

Legal
California has now passed two laws that will have an effect on how investors, health care entities and management services ...

Social Media
A prospective aesthetic patient today will have already heard, seen or read about dermal fillers in their social media feed ...


Marketing
By AestheticsPro For medical spa owners looking to reach their clients in the most...
Top Tags
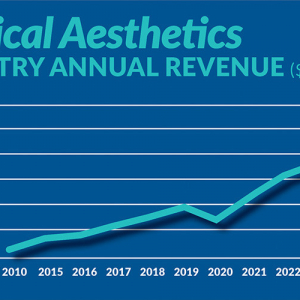
January 24, 2023
Figure displays medical aesthetics industry annual revenue ($ in billions). By Madilyn Moeller Day-to-day life...

February 24, 2021
By Bradford E. Adatto, Partner, ByrdAdattoIntravenous (IV) therapy has been used to provide nutrition and...

September 21, 2023
By Patrick O'Brien, JD, General Counsel, American Med Spa Association [AmSpa first published a...
July 19, 2023
By Patrick O'Brien, General Counsel, American Med Spa Association (AmSpa) UPDATE #2 (2/08/2024): Since...