Featured Post
Who Can Own a Medical Spa?

Legal
A newly passed law in California will prohibit certain contractual provisions between medical and dental practices and private equity groups ...

Legal
By Patrick O'Brien, General Counsel, American Med Spa Association (AmSpa)The September bulletin from the Texas Medical Board (TMB) helps to ...

AmSpa Events
Medical Spa Show 2026 is a chance for medical aesthetics professionals to reconnect with the values that brought them into ...

If there is one event in the medical aesthetics industry that stands apart as the “who’s who” gathering of leaders ...


Marketing
By AestheticsPro For medical spa owners looking to reach their clients in the most...
Top Tags
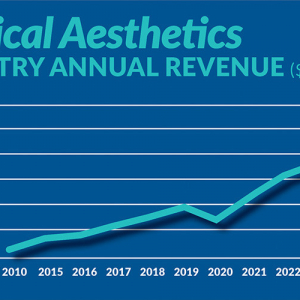
January 24, 2023
Figure displays medical aesthetics industry annual revenue ($ in billions). By Madilyn Moeller Day-to-day life...

February 24, 2021
By Bradford E. Adatto, Partner, ByrdAdattoIntravenous (IV) therapy has been used to provide nutrition and...

September 21, 2023
By Patrick O'Brien, JD, General Counsel, American Med Spa Association [AmSpa first published a...
July 19, 2023
By Patrick O'Brien, General Counsel, American Med Spa Association (AmSpa) UPDATE #2 (2/08/2024): Since...